Mechanical face seals are a complex combination of materials and design that form a system whose prime objective is maintaining the integrity of the pumping system, keeping what is inside where it belongs and preventing contamination from the outside. From the simplest design to the most complex, the system must operate across a multitude of conditions (and often beyond what the original design intended) in terms of speed, contact loads and environment. Every component in the system is a vital link contributing to the system’s success or failure. These systems enable relative motion between stationary and rotating components while simultaneously accommodating some level of axial or radial movement. The technology that has been developed and implemented in these systems has become so reliable that mechanical seals are often taken for granted. The vast majority of failures of mechanical seals can be attributed to the system in which the seal is installed, not the seal itself. One of the materials used in many mechanical seal systems is based on the fourth most common element and the basis of all life—carbon. This article will explore how this common element plays such a critical role in these complex systems. Because mechanical sealing systems must maintain tightly controlled contact between one rotating and one stationary face, interface stability must be maintained across a potentially wide spectrum of conditions. Face flatness is critical and is measured in millionths of an inch; unexpected distortion will change the interface dynamics significantly. With the proximity of the two face materials, contact is inevitable and the materials must be able to operate with some level of self-lubricity so they do not damage each other, which would create a path across that interface that enables system leakage.
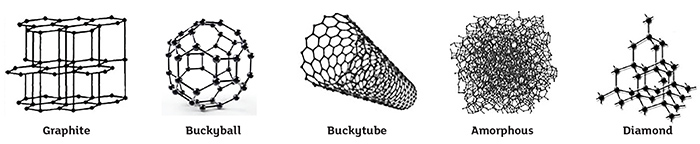 Figure 1. Five of the eight carbon allotropes. (Graphics courtesy of the author)
Figure 1. Five of the eight carbon allotropes. (Graphics courtesy of the author)- Filler (20-80 percent): Natural Graphite, Synthetic Graphite, Petroleum Coke, Lampblack
- Binder (15-50 percent): Synthetic Resins, Coal Tar Pitch, Petroleum Pitch, Metals, Carbohydrates
- Additives (0-10 percent): Film Formers, Abrasives, Antioxidants, Graphitizing Aids
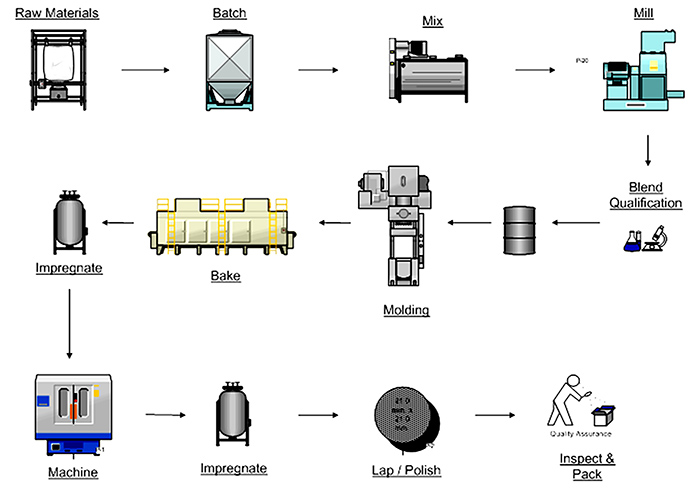 Figure 2. Mechanical carbon process overview
Figure 2. Mechanical carbon process overview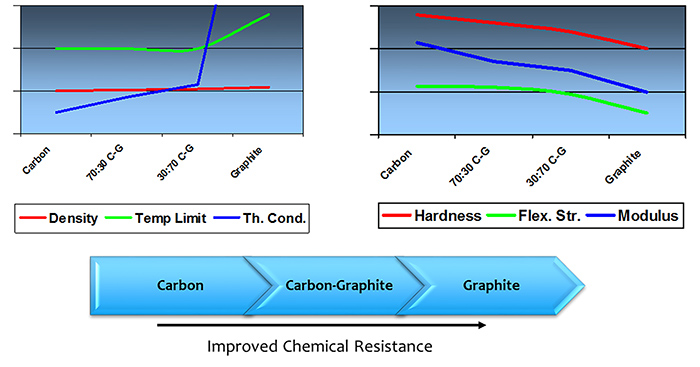 Figure 3. Effect of composition on physical, thermal and chemical properties
Figure 3. Effect of composition on physical, thermal and chemical properties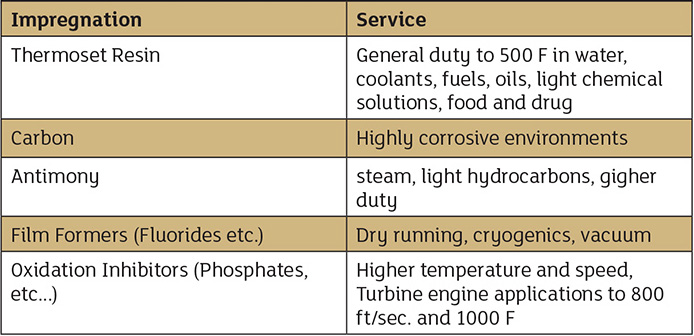 Table 1. Applications for various formulations
Table 1. Applications for various formulations
