Evaluate properties of rubber during installation & seal life.
Ergoseal Inc.
11/01/2018
Elastomers (or rubbers) are a ubiquitous family of materials whose use stretches across nearly the entire range of mechanical seal designs. From plant-sourced natural rubber, so named by John Priestly in 1770 for its utility in rubbing away pencil graphite, to petroleum-sourced synthetic rubber first developed around the turn of the 20th century, elastomers and their properties are familiar but should not be overlooked—especially when dealing with mechanical seals.
How Elastomers Work in Mechanical Seals
Rubber seals come in a variety of profiles—O-rings, cup gaskets, bellows diaphragms, sealing/wiper lips and many others. They are classified as either static or dynamic and create positive pressure against surfaces to eliminate or control the leakage of liquids and/or gases while preventing the entrance of external contaminants such as dust and dirt.1 Static sealing occurs between adjacent surfaces with no relative motion, such as between the pump casing and cover. Due to frictional wear and heat generation, dynamic sealing is less straightforward, occurring between adjacent surfaces that are continuously or intermittently moving relative to another, such as between the pump casing and shaft.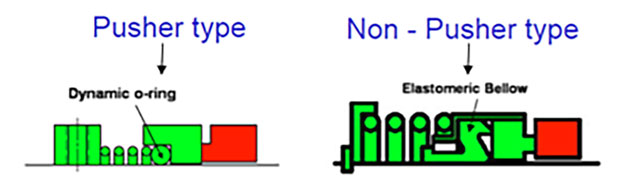 Image 1. Secondary, dynamic elastomeric seals in mechanical face seals. (Image courtesy of FSA)
Image 1. Secondary, dynamic elastomeric seals in mechanical face seals. (Image courtesy of FSA)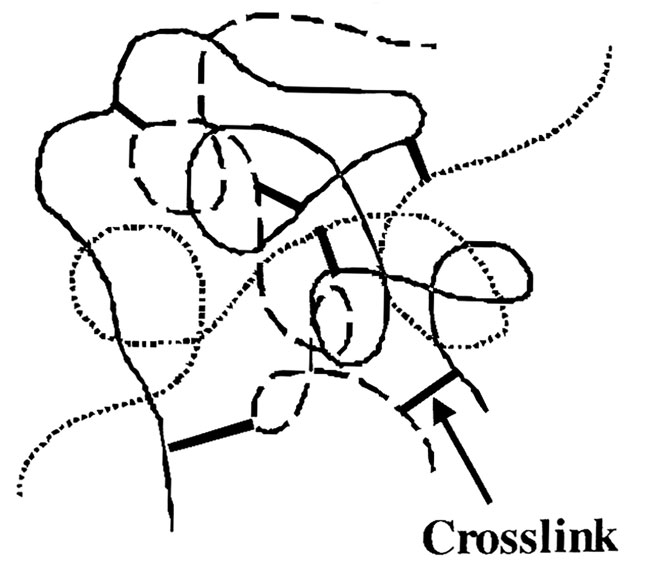 Image 2. Representation of three polymer chains after formation of crosslinking via vulcanization1 (Image courtesy of Fundamentals of Rubber Technology)
Image 2. Representation of three polymer chains after formation of crosslinking via vulcanization1 (Image courtesy of Fundamentals of Rubber Technology)Benefits of Rubber
The definition of an elastomer provides initial insight into where rubber gets its resilient sealing quality: “a macromolecular material which, in the vulcanized state and at room temperature, can be stretched repeatedly to at least twice its original length and which, upon release of the stress, will immediately return to approximately its original length.”2 When the rubber is squeezed by the adjacent surfaces of the clearance gap to be sealed, it has the characteristic properties of malleably deforming and taking the shape of each surface in response to the stress and applying a force back against the surfaces in its attempt to return to its original dimensions. Elastomers consist of large molecules called polymers (from the Greek “poly” meaning “many” and “meros” meaning “parts”), which are long chains of the same or different repeating units, called monomers, usually linked together by carbon-carbon bonds (the most notable exception being silicone elastomers, which are linked by silicon-oxygen bonds). Soft and hard plastics are also composed of polymers. However, the regularity of the monomers in their polymer chains allows neighboring segments to align and form crystals, making the macromolecular plastic material rigid and inelastic.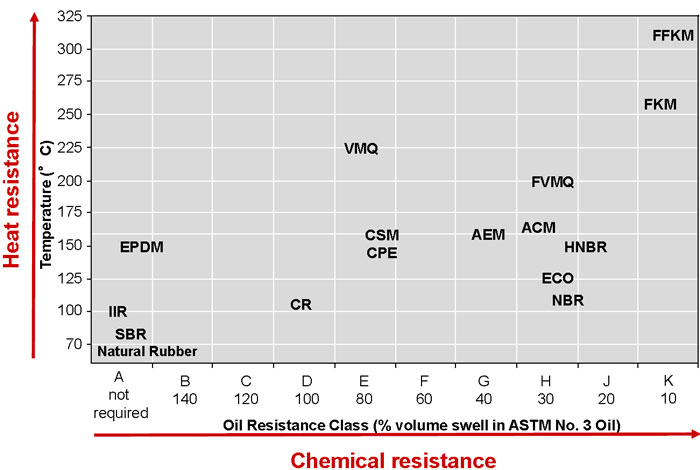 Image 3. Rubber classification by heat resistance vs. oil-swell resistance according to ASTM D2000 (Image courtesy of the author)
Image 3. Rubber classification by heat resistance vs. oil-swell resistance according to ASTM D2000 (Image courtesy of the author)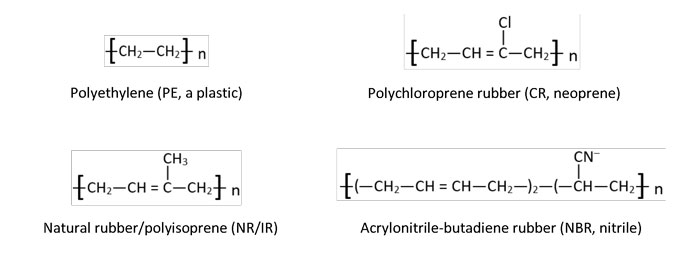 Image 4. Regular, repeating polymer chain of polyethylene plastic versus elasticity-imparting side goups –Cl, –CH3, and –CN– of neoprene, polyisoprene and nitrile rubber, respectively. The bracketed monomer units repeat a number of, or n, times.(Image courtesy of the author)
Image 4. Regular, repeating polymer chain of polyethylene plastic versus elasticity-imparting side goups –Cl, –CH3, and –CN– of neoprene, polyisoprene and nitrile rubber, respectively. The bracketed monomer units repeat a number of, or n, times.(Image courtesy of the author)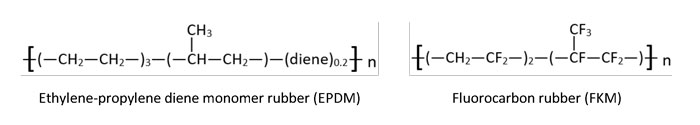 Image 5. Lack of unsaturation (i.e., no carbon double bonds) affords EPDM and FKM with better heat resistance. Hydrogen substitution with fluorine increases FKM’s stability in harsh operating conditions further still. (Image courtesy of the author)
Image 5. Lack of unsaturation (i.e., no carbon double bonds) affords EPDM and FKM with better heat resistance. Hydrogen substitution with fluorine increases FKM’s stability in harsh operating conditions further still. (Image courtesy of the author)Selecting the Right Rubber
Selection among the more than 20 families of elastomeric polymers and their contrasting properties and costs is facilitated by consulting a seal manufacturer. The process begins with an inventory of performance requirements and environmental conditions to which the rubber will be exposed. The sealed fluid chemistry, temperature range, pressure range, frequency and speed of relative motion, degree of lubrication and abrasives present, and several other interdependent design variables must be considered. Typical elastomer performance descriptors include resistance to chemical attack, such as by oil, polar solvents, acid, alkali, flame and oxygen/ozone; temperature rating (both high and low); and resistance to high temperature versus resistance to swelling in oil (per the ASTM D2000 rating/nomenclature system). Challenging design constraints more specific to an application may not be insurmountable. For example, if an unavoidable lack of lubrication for a lip seal results in premature failure due to frictional heat buildup, lubricity can be added to the rubber compounding via graphite, polytetrafluoroethylene (PTFE) or other dry lubricants. Further description of polymer structures informs rubber selection. One method for breaking up the regularity of polymer chains and hindering crystallization into rigid plastics is adding molecular branching segments or side groups to the main chain.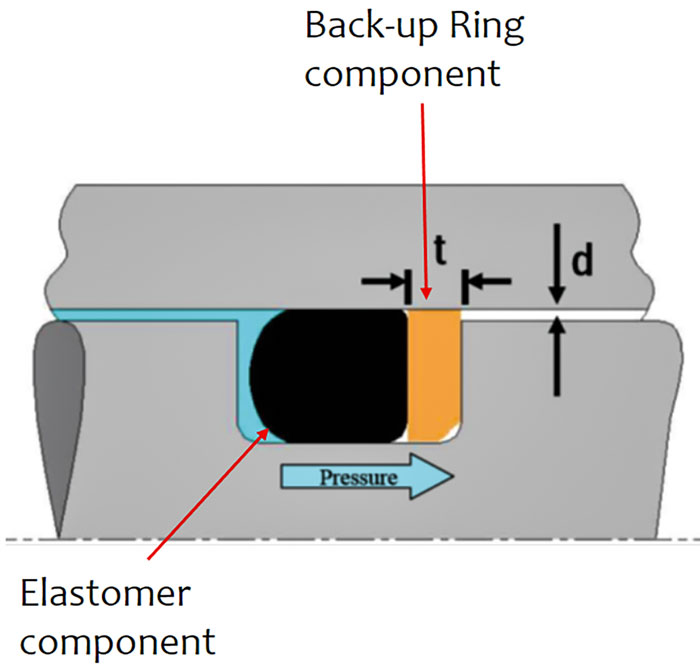 Image 6. O-ring installed in groove, with backup ring for increased pressure capability (Image courtesy of FSA)
Image 6. O-ring installed in groove, with backup ring for increased pressure capability (Image courtesy of FSA)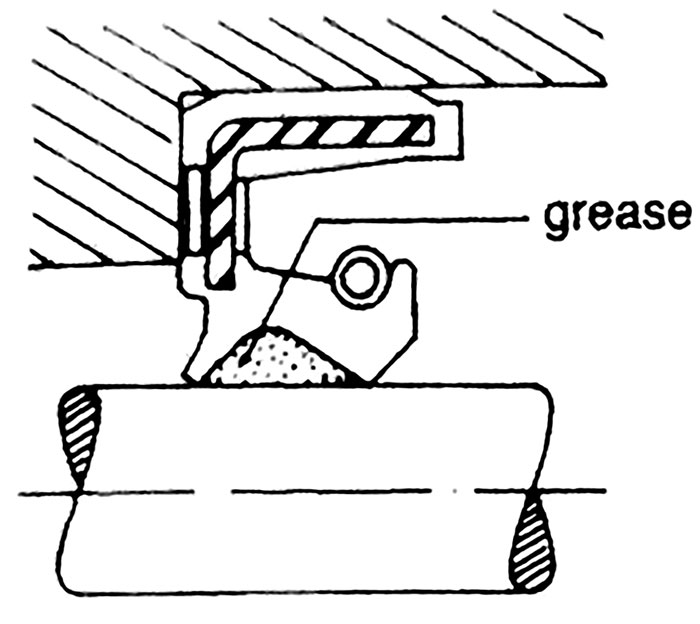 Image 7. Prelubricated lip seal properly bottomed out in bore shoulder (shaft is out of scale) (Image courtesy of the author)
Image 7. Prelubricated lip seal properly bottomed out in bore shoulder (shaft is out of scale) (Image courtesy of the author)Installation & Design Variables
Many elastomeric sealing devices are highly engineered, precision-molded products that have a service life that is dependent on proper equipment checks, handling, installation, startup and environmental controls. Depending on the seal type used, dimensional checks of groove dimensions, axial shaft movement, shaft deflection, shaft run-out, seal chamber face run-out, seal chamber bore concentricity, and other equipment features may apply. To avoid extruding rubber in high pressure applications, clearances between surfaces sealed should be minimized and/or a more rigid support member, such as a backup ring, can be installed on the low pressure side. Bore edge leads, shaft ends, splines and keyways should be checked. Rubber sealing elements should be protected from all sharp edges and tools. Especially for dynamic applications, surface finish of the appropriately corrosive-resistant shaft alloy, coating or sleeve material in the seal contact area should be free of machining imperfections and between 10 and 20 microinches (μin). Ra—rougher finishes accelerate wear and smoother finishes prevent development of a lubricating fluid film at the seal interface. When installing O-rings, lightly coat them with compatible lubricant, ensure any stretching is uniform and less than 50 percent of free diameter and avoid any twisting. When installing a lip seal, prelubricate the seal lip, use the proper installation tool in a controlled manner, bottom out the tool or seal to avoid cocking and check for parts interference.Troubleshooting Seal Failure
When analyzing failed elastomeric seals, chemical incompatibility and exposure to excessively high temperatures are associated with several similar symptoms, including hardness, cracking, extrusion and compression set. Swelling is more associated with chemical incompatibility, and extrusion may also occur due to exposure to excessive pressures. Extrusion, compression set, cuts or abrasions and mechanical damage or wear may also be signs of equipment issues, including excessively large extrusion gaps, excessive compression, sharp edges and improper surface finishes. Image 8. From left to right, failure modes of swelling, extrusion, cracking and compression set (Images courtesy of FSA)
Image 8. From left to right, failure modes of swelling, extrusion, cracking and compression set (Images courtesy of FSA)
