Mechanical seals used in food and beverage manufacturing frequently operate in indirect food contact conditions—interfaces exposed to condensate, aerosols, clean-in-place (CIP)/sterilization-in-place (SIP) media and transient product splashes. Despite the “indirect” designation, the expectations in major markets for migration-control, documentation and good manufacturing practice (GMP) often mirror those for direct food contact.
This article presents a decision framework for selecting elastomers, thermoplastics, hard faces, metals and adhesives for mechanical seals where materials may indirectly contact food, integrating current regulatory changes. It also translates these developments into material selection criteria, testing strategies and supplier documentation requirements applicable to pumps, mixers, valves and homogenizers, providing an audit-ready approach that reduces risk while supporting global commercialization.
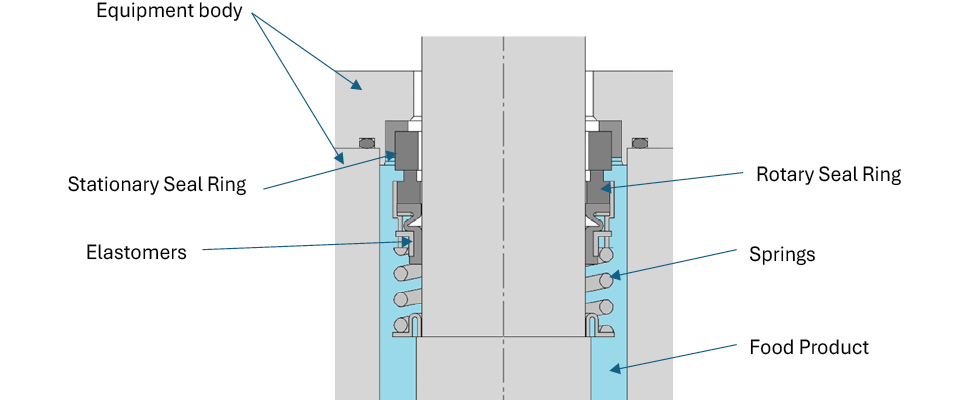
Seals at pump, mixer and valve interfaces are exposed to heat, pressure, motion and chemicals that can accelerate diffusion of residual monomers, oligomers or additives. In indirect contact scenarios, condensate, aerosols or barrier fluids can act as migration vectors. Accordingly, seal stacks (elastomers, plastics, faces, metals and adhesives) should be selected and validated as food-contact materials (FCMs) to ensure any transfer to food remains safe and compliant (Image 1).
John Crane)
Regulatory Landscape
European Union (EU): The plastics regime under Regulation (EU) No 10/2011—consolidated to March/April 2025—was amended by Regulation (EU) 2025/351, introducing a “high degree of purity” for substances and reinforcing nonintentionally added substances (NIAS) controls, new documentation and labeling for repeated-use plastics. In parallel, Commission Regulation (EU) 2024/3190 prohibits bisphenol A (BPA) and certain hazardous bisphenols/derivatives in food-contact materials effective Jan. 20, 2025, with defined transition periods.
U.S.: Title 21 Code of Federal Regulations (CFR) pathways (food additive petitions, food contact notification [FCNs], thresholds of regulation) continue to govern FCMs. The Food and Drug Administration (FDA)’s Human Foods Program (HFP) (implemented Oct. 1, 2024) centralizes oversight. In March 2025, the Department of Health and Human Services (HHS) directed FDA to pursue rulemaking to eliminate the self-affirmed “generally recognized as safe” (GRAS) pathway—moving toward mandatory FDA review and greater transparency.
China: China’s Guobiao (GB) 4806 series regulates plastics, coatings, inks and adhesives. GB 4806.15-2024, effective Feb. 8, 2025, is the first adhesive standard, defining direct vs. indirect contact adhesives, positive lists and general specifications (overall migration limit [OML], heavy metals, primary aromatic amines [PAA] for relevant chemistries), plus labeling requirements.
Japan: Japan’s Positive List system for synthetic resins reached full enforcement June 1, 2025 (transition ended May 31, 2025). Resin-based seal components must align to positive list tables with supporting documentation.
MERCOSUR and other regions: Mercado Común del Sur (MERCOSUR) follows an EU-like architecture with framework provisions and positive lists for plastics (e.g., Grupo Mercado Común [GMC] Res. 02/12, 32/07; general provisions 56/92).
Harmonization trend: Expert groups advocate alignment of risk assessment principles and analytical/testing approaches, enabling single global test plans mapped to local rules.
Use Scenario Envelope
Define food types and corresponding simulants/time-temperature schedules, characterize contact mode and cumulative time; specify thermal/pressure cycles (CIP/SIP), list cleaning/disinfection chemistries and declare destination markets. This envelope determines worst-case migration and documentation expectations across jurisdictions.
Materials for the Seal Stack
Elastomers
- Ethylene propylene diene monomer (EPDM): Strong performance in hot water/steam and alkaline CIP
- Vinyl methyl silicone (VMQ): Thermal stability but potential swelling in fats/alcohols
- Fluoroelastomer (FKM): Improved resistance to oils/solvents
- Perfluoroelastomers (FFKM): Maximum chemical/thermal resistance at higher cost
For EU trade, even if elastomers are outside plastics regulation scope, simulant-based data and EU-style GMP documentation are commonly expected. U.S. positions should reference specific CFR/FCN coverage. Bonded elastomer systems may bring adhesives into scope (GB 4806.15) for China. Purge BPA-based curatives/processing aids per the EU prohibition.
Thermoplastics and composites:
Polytetrafluoroethylene (PTFE) (virgin) is a hygienic default; filled PTFE requires additive disclosure and migration control. Polyether ether ketone (PEEK)/polyphenylene sulfide (PPS)/polyoxymethylene (POM) provide structural integrity; verify positive list status for Japan and Plastics Regulation obligations in the EU. Exercise caution with recycled plastics near product pathways given Reg. (EU) 2022/1616.
Hard faces: Silicon carbide (SiC) and alumina provide wear resistance and chemical stability; verify leachables under acidic simulants when condensate can reach food. Carbon graphite faces often use resin/oil impregnants; avoid BPA-based systems and document migration.
Metals and surface treatments: 316L stainless remains standard; avoid finishes that elevate nickel-chromium alloys (Ni/Cr) leaching in acidic media; document passivation under GMP.
Adhesives and tie-layers: Bonded lips, laminated carriers and antifretting films require adhesive scrutiny. China’s GB 4806.15 mandates composition/migration evidence with substrate context and labeling. EU requires BPA-free adhesive systems and Japan demands positive list alignment for resinous components.
Engineering for Cleanability & Low Migration
Specify hygienic surface finishes (e.g., roughness average (Ra) ≤ 0.8 micrometer (µm) in product zones), minimize crevices and control thermal transients to prevent polymer decomposition and NIAS formation. In double seals, select barrier fluids with food-contact rationales and document potential cross-contact scenarios.
Testing Strategies
- EU: Simulant-based migration per Plastics Reg. annexes and Declaration of Compliance (DoC) with GMP; include NIAS assessments per 2025 amendments.
- U.S.: Complement CFR/FCN positions with extractions; anticipate mandatory GRAS notifications.
- Japan: Ensure positive list conformance and appropriate simulants/methods.
- China: Provide GB 4806.15 adhesive datasets including PAAs where applicable.
- MERCOSUR: Map EU simulants and lists to regional conditions.
Supplier Qualification & Documentation
Expect market-specific declarations of compliance (DoCs); migration/extraction reports aligned to use envelope; GMP evidence and change-notification clauses referencing the EU BPA prohibition and plastics amendments. Watch for vague “FDA compliant” boilerplates without CFR citations and adhesive black boxes for China supply.
Application Examples
- Dairy pump: SiC-SiC faces; EPDM secondary seals with EU-style data; bonded gaskets need adhesive compliance
- Tomato sauce mixer: Alumina-carbon faces; FKM seals; BPA-free coatings/adhesives
- Brewery transfer: SiC-carbon; methyl vinyl silicone (VMQ) vs. FKM based on swelling; Japan positive list confirmations
Portfolio Controls: Restricted Chemistry & NIAS
Implement a BPA exit plan across coatings/adhesives; establish NIAS screening and toxicological triage consistent with EU purity thresholds; monitor California Prop 65 bisphenol developments for U.S. distribution.
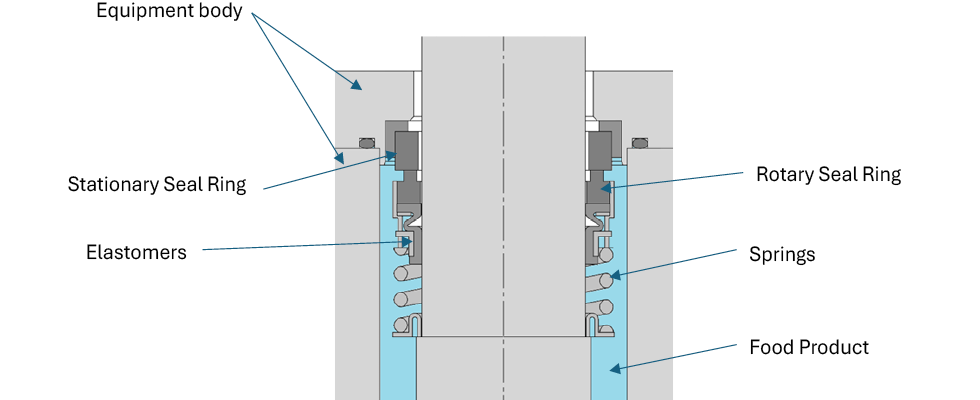
Treat indirect-contact seals as regulated FCM assemblies. Prioritize precise use envelopes, FCM-ready materials, a global master test plan and robust supplier change control. With EU plastics amendments and BPA prohibition, FDA HFP and GRAS reform, China GB 4806.15 and Japan’s positive list enforcement, requirements are rising—but pathways to compliance are clearer (Image 2).
This guidance integrates cross-regional regulatory developments and harmonization efforts, with contextual benchmarks drawn from prior internal comparative analyses of global FCM frameworks.
We invite your suggestions for article topics as well as questions on sealing issues so we can better respond to the needs of the industry. Please direct your suggestions and questions to sealingsensequestions@fluidsealing.com.
References
- Access2Markets. (2025, March 7). EU prohibition on the use and trade of Bisphenol A from 20 January 2025. European Commission.
- Atsumi & Sakai. (2025, May 30). Japan’s Positive List System for utensils, containers and packaging intended for food use (Effective from June 1, 2025).
- Eur-Lex. (2025). Commission Regulation (EU) No 10/2011 on plastic materials and articles intended to come into contact with food (consolidated).
- Eur-Lex. (2025). Commission Regulation (EU) 2022/1616 on recycled plastic materials and articles intended to come into contact with foods (consolidated).
- Food Packaging Forum. (2025, April 28). EU plastic FCM regulations amended (Regulation [EU] 2025/351).
- Food Safety News. (2024, September 25). FDA’s new Human Foods Program kicks off on Oct. 1.
- Haynes and Boone, LLP. (2024, June 4). FDA obtains approval for massive reorganization: What it means for the food industry.
- HHS. (2025, March 10). HHS Secretary directs FDA to explore rulemaking to eliminate self-affirmed GRAS.
- Hogan Lovells. (2025, January 27). EU bans BPA in food contact materials: What does it mean for industry?
- ILSI Europe. (2025, April 8). Global experts call for harmonised safety assessments of food contact materials.
- Intertek. (2025, April 14). EU approves Regulation (EU) 2025/351 amending plastic FCMs, recycled plastics and GMP.
- Intertek. (2025, January 7). EU – Regulations on the use of BPA in food contact materials and products.
- Keller and Heckman LLP. (2024, March 15). China announces the standard for food-contact adhesives (GB 4806.15-2024).
- MHLW / Food Packaging Forum. (2024,
June 25). Japan preparing food contact material regulation for June 2025 enforcement deadline. - packaginglaw.com. (2023, December 13). Japan publishes final version of the Positive List.
- packaginglaw.com. (2025, December 3). FDA’s proposed GRAS reform rule under OMB review.
- QIMA. (2025, February 22). EU adopts ban of Bisphenol A (BPA) in food contact materials and articles.
- SGS Digicomply. (2024, October 9). China’s new standard for adhesives: GB 4806.15-2024 (effective Feb 2025).
- SGS SafeGuardS. (2025, March 4). EU revises FCM regulations: Regulation (EU) 2025/351.
- SGS SafeGuardS. (2025, January 16). EU regulates BPA and other bisphenols in FCMs: Commission Regulation (EU) 2024/3190.
- Sun Chemical. (2022). Legislation – Food packaging: MERCOSUR (overview of GMC Resolutions).
- U.S. FDA. (2024, May 30). FDA’s reorganization approved for establishing unified Human Foods Program.
- U.S. FDA. (2024, Oct. 1). FDA modernization efforts: Human Foods Program and field operations.
- Finnish Food Authority. (2025). Plastic Regulation (EU) 10/2011—latest update including 2025/351.
- Finnish Food Authority. (2025). Regulation on recycled plastic (EU) 2022/1616—overview.
- SGS. (2024, May 13). China announces two standards for food contact materials (incl. GB 4806.15-2024).
- SGS. (n.d.). Food contact material regulations – MERCOSUR region.
- Lee, D. (2026). 2026 Global FCM Regulations (Internal presentation, John Crane).
For more Sealing Sense articles, visit pumpsandsystems.com/tags/sealing-sense.

