These technologies allow membrane-based distillation components to be used to produce high-purity drinking water from heavily compromised water sources.
09/08/2015
Recent developments in the field of nanotechnology have resulted in a positive impact on two of the world's largest industries—energy and water. These advancements have allowed scientists and engineers to develop new tools that work with pumping systems to improve and streamline the wastewater treatment process. Some believe that the commercial use of nanotechnology in key applications will help meet the worldwide drive to reduce carbon emissions and redefine the energy-water nexus.
 Image 1. Nanostructured polymer membrane (Images and graphics courtesy of Dais Analytic)
Image 1. Nanostructured polymer membrane (Images and graphics courtesy of Dais Analytic)How Nanomaterials Work
Nanotechnology is broadly defined as science, engineering and technology that is limited to a length scale of 1 to 100 nanometers.1 One such material is a family of nanomaterials created by the chemical modification of styrene block copolymers that are widely used in glues and gaskets. Modified copolymers can be used to produce innovative membranes that provide molecular pathways for the transport of water between the surfaces without traditional porosity. The block copolymer starting resin contains discrete blocks of styrene that are interspersed in various patterns and blocks of olefin polymers that are inherently hydrophobic and provide elasticity to the polymer. A variety of proprietary processes are used to covalently bond an acidic functional group to the styrene blocks of the copolymer without modifying the hydrophobic regions. The acid group is extremely polar, which makes that region of the polymer hydrophilic. When a liquid solution of the polymer is cast onto a flat surface and the solvents evaporate away, the hydrophobic and hydrophilic regions of the polymer exhibit different wetting characteristics. This process causes self-organization of the polymer as the different blocks attempt to minimize or maximize their interaction with the remaining solvent. Upon solidification, the polymer layer contains a pattern of hydrophobic and hydrophilic regions that are 5 to 30 nanometer (nm) in diameter, making the membrane a nanostructured material. These hydrophilic regions swell with water molecules that force the polar functional groups further from each other, a spontaneous change that reduces the electrostatic repulsion forces between the functional groups. These regions are contiguous with the neighboring hydrophilic regions, connecting one surface of the membrane to the opposite surface with a continuous chain of water molecules. The hydrophobic regions of the material provide a strong, flexible structure that keeps the material from dissolving. The polymer membrane is sensitive to any differential in vapor pressure between the two surfaces. As water molecules evaporate from one surface of the membrane, they create local imbalances in the electrostatic interactions between functional groups and water molecules, resulting in the redistribution of water molecules to replace the evaporated molecules. Image 2. Transmission Electron Micrograph (TEM) showing hydrophilic and hydrophobic regions within the polymer.
Image 2. Transmission Electron Micrograph (TEM) showing hydrophilic and hydrophobic regions within the polymer.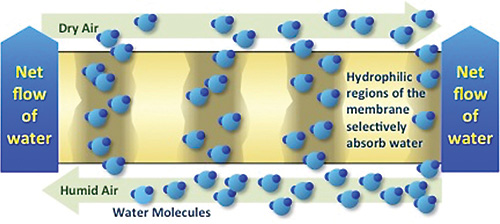 Figure 1. Nanomaterial operation
Figure 1. Nanomaterial operation- selectively move water from one face to the other at a higher rate than nitrogen, oxygen and other gases, as well as dissolved ions and materials.
- rapidly transport water molecules between surfaces to allow a high flux rate of water molecules per unit of area.
- demonstrate durability in a variety of environments.
- use commercially available methods to achieve high-volume production.
- http://www.nano.gov/nanotech-101/what/definition
- https://www.pumpsandsystems.com/news/2015-05-20/drought-driving-greater-reliance-wastewater-reuse-california#sthash.q6UwEnrh.dpuf
- Throughout this document, the terms ppm, parts per million, and mg/L are used interchangeably.

