The ancient Greek philosopher Plato is credited with coining the phrase, “Necessity is the mother of invention,” meaning that a need or problem encourages creative efforts to meet the need or solve the problem. It is unknown whether that phrase was going through Jim Wilden’s head as he watched water from a ruptured pipe gush into a shop at a steel factory in San Bernardino County, Calif., 50 years ago. Knowing that it needed to be removed, Wilden went to work, and in 1955, he had the solution—the air-operated double-diaphragm (AODD) pump.
The same ingenuity still occurs today, but now for a multitude of biopharmaceutical processes. The AODD pump has evolved to uniquely solve complex fluid transfer needs in this industry. Because of the critical nature of some processes, inefficient product transfer methods such as manual rolling carts with containers, purging tanks to evacuate product or manual gravity feeding transfers have been common. However, specifically designed AODD pumps for this industry now allow further use of pumped transfer processes (and associated production and energy efficiency) approaching the degree already found in the general chemical/industrial sector.
First, Do No Harm
The diaphragm pump already has a cousin in the industry…the diaphragm valve. The diaphragm valve has long been the valve of choice in these types of applications because of its high product containment and clean ability traits. These are also available with AODD pump technology, along with a sealless stem and shaft-free product-side environment.
This is important as both the diaphragm valve and diaphragm pump have less risk of producing product damaging shear, and neither technology has dynamic seals that would risk leaks that could contaminate the product or the production environment.
So with innovations and enhancements for the ultra-sanitary conditions needed by the pharmaceutical industry, the diaphragm pump is now an attractive option for many fluid transfer needs. These processes—and the products they produce—must meet a wide array of regulations and certifications to ensure that they are being performed in a high-purity environment. Among the regulations that AODD pumps can satisfy are those from EHEDG, 3A, CE, ATEX, USP Class VI and FDA CFR 21.177. This includes a validation package with mill, 3.1b, polish, passivation and classified area use certifications.
The liquids can run the gamut from extremely shear-sensitive to extremely viscous, and semi-solids can range from liquid glucose to polymer slurries. Pharmaceutical and biochemical fluids currently pumped with diaphragm pumps include: blood and by-products, live cell cultures and vaccine producing solutions, egg emulsions for vaccine production, pill coatings, eye care solutions, fluids for oncology, specialized disinfectants, nutraceuticals, vitamins, topicals (creams/lotions) and filter media. The use of AODD technology can guarantee safe transfer during the production process.
According to Hoover’s, Inc., which analyzes companies and industries that drive the economy, as many as 1,500 companies in the U.S. manufacture and market pharmaceuticals (defined as a compound manufactured for use as a medicinal drug), with combined annual revenue of more than $200 billion. These numbers indicate that the manufacture of pharmaceuticals is one of the lynchpins of the American economy. The actual creation of pharmaceuticals involves one of three major methods:
Synthesis—using chemical reactions to build a drug from simpler components
Extraction—using solvents to remove and purify a drug from a natural source
Biotechnology—using methods such as gene-splicing or the production of antibodies using mammalian (animal-based) cells
No matter the method used to produce biopharmaceuticals, the actual manufacturing process is a precise one that must be performed under demanding, exacting conditions, often in a cleanroom environment that prohibits instances of product leakage, fouling or cross-contamination.
AODD Benefits
Specifically, AODD technology is a boon to pharmaceutical manufacture in a number of crucial areas:
Sterile product transfer—AODD pumps remove the need for gas-purge systems in continuous processes because the technology allows both the filling and emptying process to occur at the same time while keeping the product contained and pure.
Process flexibility—AODD pumps can handle highly variable process conditions found in many hygienic applications.
Sampling—complex pharmaceutical processes under strict conditions require frequent and multipoint sampling. AODD pumps provide the ability to extract these samples while maintaining a high degree of containment and avoiding cross-contamination.
Clean in place (CIP)—the AODD pump’s self-priming, dry-running and sealless design is ideal for CIP operations.
Chemical feed—this is a traditional role for AODD pumps as their sealless design and reliable product containment ensure safety when handling volatile or potent chemicals.
Ingredient unloading—because they self-prime, run dry and have negative suction lift, AODD pumps meet pharmaceutical-validation requirements and can be applied where needed.
Product recovery and semi-solids removal—again, the AODD pump’s dry-run, self-priming and full product containment makes it ideal for use in most filtering or separation processes.
Chromatography, separation, purification and filter feed—these processes often require shear-sensitive transfer and constant pressure feed, traits found in AODD pumps. Extracting delicate cell structures from centrifuge discharge is a good example (see Figure 1)
Maintenance
Simplicity defines the cleaning and maintaining of these pumps. In the highest hygienic configurations, these pumps are designed for CIP so that manual labor and contamination risks do not occur. However, at the same time, these pumps have been purposely designed to be simple to disassemble for either manual cleaning procedures or routine maintenance.
Power and Fluid End Separation
As biopharmaceutical processes evolve, more attention is paid to optimizing the process. The separation of mechanical and utility functions is an example of area/floor space optimization. AODD pumps can be split so that the “power side” is mostly located remotely in an unclassified area and the fluid end can be placed in a classified area, with only an instrument air-supply line connecting the two. This means that electric motors, oil-filled gear cases and greased bearings no longer need to be located in the clean area, raising the level of hygiene and reducing the risk of product contamination.

Integral Piston Diaphragm
For high purity industries, one primary innovation that has made this pump among the most viable selections in the industry has been the integral piston diaphragm (IPD) (see Figure 1). Unlike traditional pump diaphragms that have an outer plate that supports the diaphragm that is subject to more difficult cleaning or can be a potential leak point, the IPD is completely laminated with USP Class VI PTFE on the product contact side. This offers the highest degree of containment and clean ability among pumps.
Figure 1. Integral piston diaphragms (IPD) are featured in certain AODD pumps.
Inside an AODD Pump
The uncomplicated design of AODD pumps features few moving parts, and those that do move have simple, specific tasks:
Air chamber—houses the air that powers the diaphragms
Air distribution system—the heart of the pump, it is the mechanism that shifts the pump to create suction and discharge strokes
Outer diaphragm piston—connects the diaphragms to the reciprocating common shaft and seals the liquid side from the air side of the diaphragm
Inner diaphragm piston—located on the air side of the pump, it does not come in contact with the process fluid
Valve ball—seals and releases on the check-valve seats, allowing for discharge and suction of process fluids to occur
Valve seat—provides the ball valves a place to check
Discharge manifold—allows fluid to exit the pump through the discharge port, which is typically located at the top of the pump
Liquid chamber—separated from the compressed air by the diaphragms, it fills with process fluid during the suction stroke and is emptied during the discharge stroke
Diaphragm—acts as a separation membrane between the process fluid and the compressed air that is the driving force of the pump; to perform adequately, diaphragms should be of sufficient thickness and of appropriate material to prevent degradation or permeation in specific process-fluid applications
Inlet manifold—allows fluid to enter the pump through the intake port located at the bottom of the pump
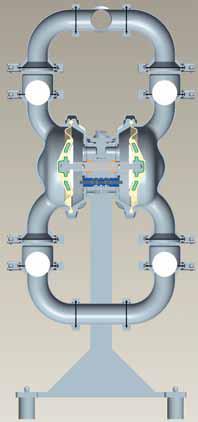
(Above): Inside an AODD Pump

