
The food and beverage industry is one of the oldest industries, and as such, it is also one of the most innovative and diverse. It includes agriculture, food processing, manufacturing, transportation and servicing. The public unknowingly interacts with these groups daily by buying packaged food at the grocery store or dispensing cola from a soda fountain machine.

The average person does not give much thought to the materials used in the lining of a food container or the tubing in a soda machine. So, how do users know they are safe? Rest assured, there are several requirements, established by agencies, to ensure the food and beverages will not make people sick.
Manufacturers must select the food contact material that is compatible with their food category to reduce the risk of contamination. Food contact materials are any materials that have foreseeable direct or indirect contact with food or potable beverages. For example, the primary function of the tubing on a soda machine is to transport fluids while preventing odors, microorganisms and pests from contaminating the carbonated drinks. However, chemicals might still transfer from inside the tubing to the beverage. This migration of chemicals, otherwise known as leaching, impacts food safety and quality.
Foods are classified into five categories: aqueous, acidic, alcoholic, fatty and dry. Migration can occur in any of these categories in the following ways:
- Acidic foods tend to be corrosive to metals.
- Fats and oils swell and leach plastics.
- Aqueous beverages disintegrate unprotected paper and cardboard.
This migration of chemicals, like virtually all chemical and physical processes, is further accelerated by heat. Migration may also be expedited with an increase of time, thickness of the material and chemical concentration.
Choosing the correct material for an application requires extensive testing. It should come as no surprise that these materials used in the food industry are heavily regulated by state, federal and international legislation. Within the United States, one of the most common regulators is the Food and Drug Administration (FDA). The FDA is responsible for protecting and promoting public health. It was not until 1958 that the FDA was granted authority by the U.S. congress to regulate food additives through the Food Additives Amendment. Receiving FDA compliance for food additives happens in three ways: existing, new and exempt materials.
Title 21 of the Code of Federal Regulations (CFR), parts 170-199, provide general and specific regulations on already existing food contact materials. Extractive limitations are also provided for each material.

A manufacturer must register any new materials that are not already existing. This requires a food contact notification (FCN). The more common and less lengthy process is the food additive petition (FAP).
The threshold of regulation (TOR) submission process is used for exemption of the food contact material, due to its negligible health risk.
It is the responsibility of the manufacturer of the product to take the proper actions to claim food-grade compliance.
When dealing with potable water additives, the name National Sanitation Foundation (NSF) is not far behind. NSF International is an independent, nonprofit, agency that sets standards on drinking water. Often tied with the American National Standards Institute (ANSI), its certifications require testing from NSF-accredited third-party organizations to receive approval.
One of the most stringent agencies lies within the medical industry. The U.S. Pharmacopeia (USP) has six classes of certifications. USP class VI, the most rigorous, requires compounds to be made from ingredients with clear histories of biocompatibility that meet tighter requirements for leachates.
To stay within compliance, food processing plants will ask for certificates with their material orders. A favored material that is often found within the industry is polytetrafluoroethylene (PTFE). Foods are chemically aggressive and may strongly interact with the materials they touch.
PTFE is the one of the most chemically inert, nontoxic and nonflammable substances that repels debris and liquids, has a low coefficient of friction and is thermally stable, even at cryogenic temperatures. PTFE also does not absorb water and contacting dairy or moisture filled foods will not compromise their taste.
These characteristics of PTFE are advantages in sealing. Specifically, as a braided packing, thin filaments of PTFE yarn are braided into a lattice structure, cut into rings and are compressed into a stuffing box. Due to PTFE’s low coefficient of friction, the packing is used with or without a lubricant. The lubricant, made of white paraffin mineral oil, is a mixture of liquid hydrocarbons. Paraffinic and naphthenic in nature (obtained from petroleum) meets FDA and NSF registration guidelines. However, some end users prefer PTFE dry to eliminate any risk of contamination with their process fluid.
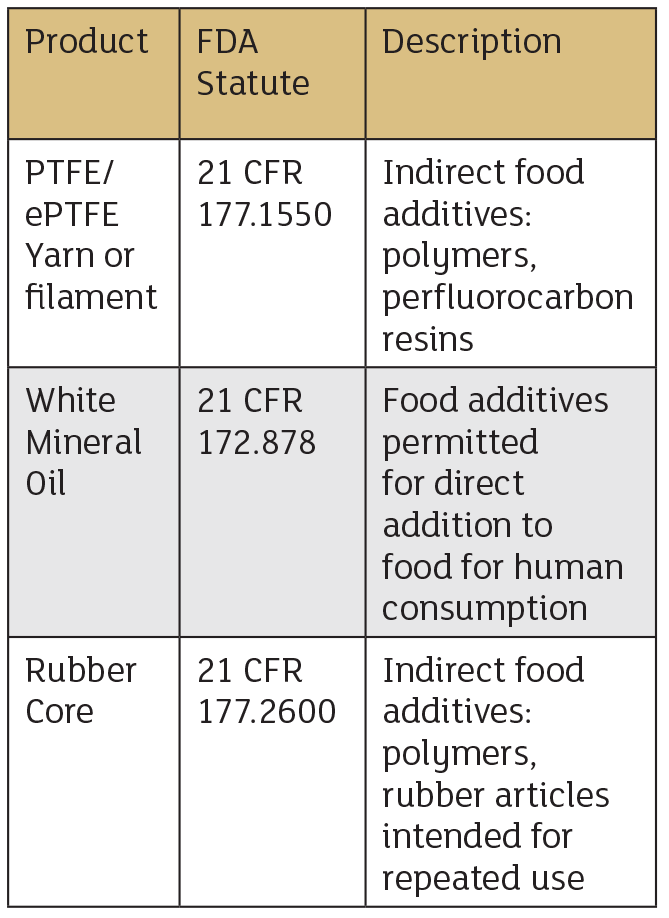
Mixers and agitators are commonly used to combine food ingredients and can be a challenge to seal due to high shaft runout. If a shaft over compresses packing in the radial direction, it can affect its sealing ability and cause premature failure. Braiding packing around an elastomer core gives the packing memory and allows it to withstand the radial shaft movement. Many elastomers are FDA compliant, including Buna-N (nitrile) and silicone.
Although all the ingredients presented with their FDA statute in the table above are food grade compliant, good manufacturing practice (GMP) should always be implemented to minimize risks of contamination. The GMP system ensures that products are consistently produced and controlled according to quality standards. For example, if the FDA-compliant PTFE filament is braided in the same braider that had a material that was non-FDA compliant, then contamination has occurred. Other risks include mislabeling the container it is placed in or handling the material without the proper equipment.
Contamination may also occur onsite at the food processing plant. Installing packing in a dry or abrasive application often includes a flush system. Flush is used to lubricate the packing rings or acts as a high-pressure barrier that prevents solids from entering the stuffing box. Water is a common flush media. When used in a food processing context, the plant must ensure that the water is properly filtered. Water filtration removes sediments, chemicals, minerals and organic matter, which can be dangerous for human consumption.
Unfortunately, there is always still a chance that adulterated food and beverage will reach the public. An unmaintained seal degrades over time and may end up in the food products. Once this occurs, the appropriate strategy is to separate the food from the user. Companies can voluntarily announce a recall to keep a reputation that shows concern for their users. The FDA also has the power to initiate that recall. This is undoubtedly a process that processing plants want to avoid at all costs.
Food products already go through an X-ray or metal detector to identify any foreign particles that may have tainted the product, but polymer seals are often missed. One way to tackle this problem is by using metal detectable plastic. The solution entails adding tiny fragments of metal powder or high-density additives into the plastic components. Doing so makes them easier to detect before leaving the facility.
There are various food contact materials used throughout the food and beverage industry, which means there are also many situations where chemical migration can occur. Manufacturers and food processors must follow their local agency standards to ensure that their products are never compromised, even in the most complex applications. Gathering the correct compliance, choosing a compatible material and taking the appropriate precautions are critical to the health and safety of consumers.
We invite your suggestions for article topics as well as questions on sealing issues so we can better respond to the needs of the industry. Please direct your suggestions and questions to sealingsensequestions@fluidsealing.com.

