Galvanic corrosion is an electrochemical process that occurs between two dissimilar metals, or between a metal and a conductive non-metallic material, when both are exposed to an electrically conductive media.
In the case of a packing gland, it occurs between a metal component and the carbon or graphite packing. Under these conditions, the material that is closest to the anodic end of the galvanic scale will be corroded in preference to the one that is closest to the cathodic end of the scale. (See Table 1.) As the distance between materials on the galvanic scale increases, a corresponding rise occurs in the rate and the extent of the corrosion.
In a valve or a pump using packing made of either graphite or carbon, a galvanic reaction may be initiated as soon as any electrically conductive fluid, such as water, is introduced. Since graphite is more cathodic than the metals that make up valves and pumps, it is the metal that may be subject to corrosive attack.
Liquid Phase Needed
Even though a valve or pump may be packed with a graphite or carbon packing, many cases exist in which the metal parts will not be subjected to galvanic corrosion. For example, an electrically conductive fluid in a liquid state must be present for the galvanic reaction to take place. The temperature of a superheated steam valve prevents the accumulation of any significant amount of water, thereby nullifying the possibility of galvanic corrosion.
Stainless Steels
Another example of when galvanic corrosion protection may not be necessary is when the equipment is constructed of austenitic stainless steels (e.g., 300 series, 630, etc.). These stainless steels are much more resistant to galvanic attack.
On the other hand, the martensitic stainless steels (e.g., 400 series) are highly susceptible to galvanic attack. If a valve or pump is constructed of martensitic stainless steel and if it will be exposed to an electrically conductive fluid for any period of time, then consideration should be given to incorporating a galvanic corrosion inhibitor system into the carbon or graphite packing sets used to seal it.
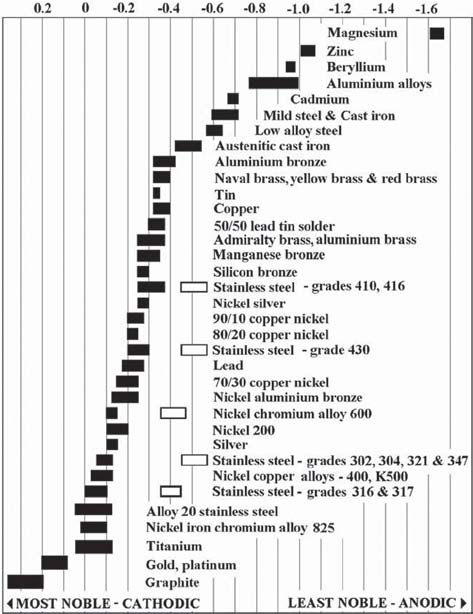
Table 1. Galvanic series
When Are Corrosion Inhibitors Needed?
The following are several examples in which corrosion inhibitors would be recommended to prevent corrosion in equipment made of less resistant alloys such as martensitic stainless steel:
- Valves and pumps that are hydrostatically tested and then stored in a wetted condition
- Steam service valves that will undergo cold downtime periods because, depending upon the valve's position within the piping system, some may be exposed to water or condensate during the cold exposure period
- Equipment that is located in an outdoor environment in which rain water can collect in the top of the stuffing box between the gland follower and the stem
- Valves, such as fire mains, that are under constant water exposure due to their intended service conditions
Corrosion Inhibitors
Two galvanic corrosion inhibitor systems are commonly available for use in compression packing. They have a common goal of nullifying the effects of galvanic corrosion. However, they are based upon two different protection mechanisms.
Active Inhibitor
Active, "sacrificial," inhibitor systems are based on the principle that the galvanic reaction will preferentially attack the material that is most anodic. As long as this most anodic material remains in contact with the conductive fluid, it will be attacked and corroded in preference to the other metals or materials present in the stuffing box. The active inhibitor systems provide, as part of the packing set, a metal of a more anodic nature than that of the material used in the construction of the valve.
The most common active inhibitor system used today is zinc powder. Being located near the extreme anodic end of the galvanic scale, zinc is more anodic than the metals that are commonly used in valves and pumps.
A zinc powder coating can be easily applied to the surface of many different types of compression packing products including die-formed flexible graphite rings and nearly any type of braided carbon or graphite. When zinc powder is applied to the surface of a packing material, it appears as a light grey coating.
Care should be excercised in the case of equipment that may see service temperatures that exceed the 419-degree-C (786-degree-F) melting point of zinc. At temperatures above its melting point, zinc may contribute to the liquid metal embrittlement of the base metal.
Passive Inhibitor
Passive corrosion inhibitor systems protect the metal surfaces of equipment by forming a barrier film on the more anodic metal component. The graphite packing and the metal surfaces must be exposed to each other through an electrolytic fluid medium for a galvanic corrosion reaction to occur. The passive inhibitor creates a continuous thin barrier film through which the galvanic reaction cannot occur.
Several manufacturers make grades of flexible graphite that incorporate phosphorus as a passive corrosion inhibitor. These inhibited materials can be used in many forms for both compression packing and gasketing products. The phosphorus is dispersed throughout the material during processing of the flexible graphite sheet from which the packing is fabricated. It is not just on the surface of the packing.
Phosphorus based corrsion inhibitors can be used for applications in which zinc may not be allowed, such as high temperature services above its melting point or cases in which zinc may not be compatible with the process fluid.
Advantages and Disadvantages
While the presence of zinc powder inhibitor can be confirmed visually, uniform dispersion can be a challenge. Unlike the gray surface of zinc powder inhibited packing materials, phosphorus is not visible. This is both a benefit and a disadvantage. Handling zinc coated packing rings will sometimes leave the installer with a residue on his hands, but the visibility of the zinc readily identifies that the protection system is in place. The invisibility of the passive phosphorus protection systems on packing rings is definitely cleaner from a handling standpoint, but the only way to tell if the phosphorus is in place is through chemical analysis.
Another benefit of using phosphorus is that it not only acts as a corrosion inhibitor but also as an oxidation inhibitor, increasing the material's ability to withstand very high temperatures without losing significant mass to oxidation.
Conclusion
By understanding the conditions under which galvanic corrosion can occur and by using readily available compression packing products with appropriate corrosion protection, galvanic corrosion is a problem that can be eliminated from your packing gland.
Next Month: How does thermal expansion influence application of PTFE packing?
Pumps & Systems, August 2011
The following members of the Compression Packing division sponsor this "Sealing Sense":
- A. W. Chesterton Co.
- Daikin America, Inc.
- DuPont Performance Elastomers L.L.C.
- Empak Spirotallic Mexicana SA de CV
- Garlock Sealing Technologies
- W.L. Gore & Associates, Inc.
- GrafTech International Holdings, Inc.
- Palmetto, Inc.
- John Crane
- Lenzing Plastics GmbH
- Nippon Pillar Corporation of America
- SEPCO - Sealing Equipment Products Co.
- SGL Technic Polycarbon Division
- Slade, Inc.
- Teadit International
- YMT/Inertech, Inc.

